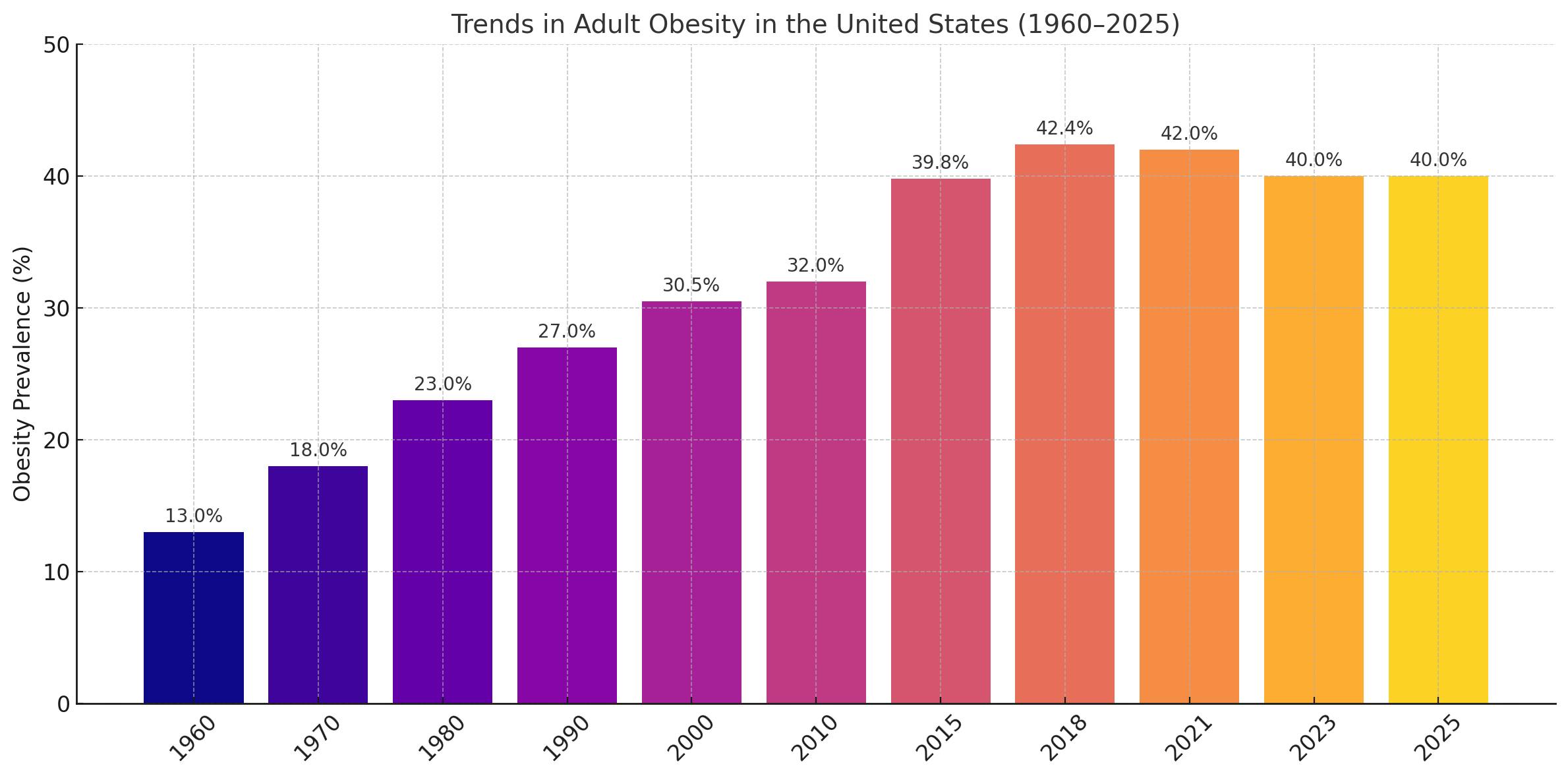
GLP‑1 receptor agonists — including Ozempic, Wegovy, Mounjaro, and Zepbound — have had a transformative impact on obesity management in the United States. Following a threefold increase in adult obesity rates from 1960 to 2021, recent U.S. health data now shows a modest but significant 2% decline from 2022 to 2025, a shift widely attributed to the rising use of GLP‑1 medications.
The adoption of GLP‑1 drugs has accelerated rapidly. Between 2019 and 2024, prescriptions increased by nearly 587%, rising from 0.3% to 2.05% of overweight or obese adults. Usage has climbed particularly among women approaching menopause, with 18.6% of women on GLP‑1s using them for weight loss compared to 9.3% of men.
Adherence rates are also improving. Nearly 63% of patients who began treatment with Wegovy or Zepbound in early 2024 remained on therapy after one year — a significant increase from 40% in 2023. Clinical trials report average weight reductions of 15–21% with semaglutide or tirzepatide over 12 months, outcomes comparable to some bariatric procedures.
As a result, the volume of bariatric surgeries has declined, reflecting a shift toward medical rather than surgical obesity interventions. However, this may prove to be a temporary trend, depending on long-term efficacy, cost, and clinical practice guidelines for GLP‑1 use.
Beyond weight loss, GLP‑1 therapies offer broader health benefits. These agents improve metabolic function, reduce systemic inflammation, and lower cardiovascular risk. Notably, the SELECT trial found a 19% reduction in all-cause mortality and a 20% reduction in cardiovascular deaths among patients treated with semaglutide.
Despite these advances, significant barriers remain. Fewer than 3–4% of eligible obese adults currently receive GLP‑1 prescriptions, due to disparities in insurance coverage, cost, and geographic access. Out-of-pocket costs often exceed $1,000 per month, prompting some individuals to seek unregulated sources, which pose safety risks.
Side effects are also a consideration. Common adverse effects include nausea, vomiting, and gastrointestinal discomfort. Long-term safety is still under review, particularly regarding risks of medullary thyroid cancer, gallbladder disease, and diabetic retinopathy. For this reason, baseline screening and ongoing monitoring are essential, especially in patients with diabetes or pre-existing thyroid conditions.
GLP‑1 receptor agonists represent the first pharmacologic intervention with the potential to reverse national obesity trends. While challenges in access, cost, and long-term safety remain, these medications are already reshaping the landscape of obesity treatment in the United States — with implications for individual health and public policy for years to come.